Advancing Health Through Clinical Trials for Better Outcomes

Call Us On

Our Location

Opening Hours
Saturday: 9:00am – 6:00pm
Sunday: Closed
Empowering Clinical Trials with Accurate and Reliable Assessments
At Selin Clinical Assessments, our mission is to provide precise and reliable evaluations for clinical trials, helping to advance the future of medicine. We believe that every trial contributes to improving lives and unlocking new possibilities for patient care. With our thorough approach, we aim to deliver accurate assessments that support groundbreaking research and medical advancements.
Our team consists of dedicated professionals with years of experience in the clinical trial industry. We work closely with researchers and organizations to ensure that each assessment is handled with care, accuracy, and attention to detail. By utilizing the latest technology and best practices, we maintain a commitment to quality and trust throughout every step of the process.
At Selin Clinical Assessments, we understand that clinical trials are critical to the development of new treatments and therapies. That’s why we prioritize efficiency without compromising on the integrity of the data we provide. Our goal is to offer seamless support, enabling research teams to focus on their core work while we handle the essential assessments.
We are proud to be a trusted partner in the clinical trial landscape, serving both local and global research initiatives. Whether you’re conducting a small-scale study or a large international trial, we are here to help ensure your research is backed by accurate and dependable assessments.
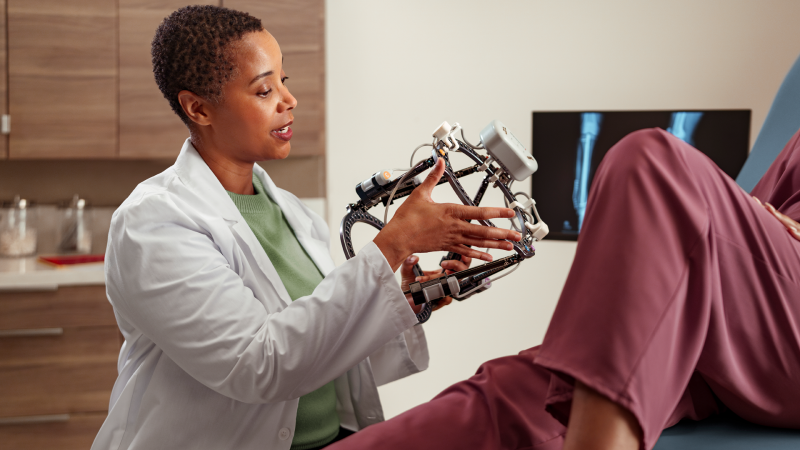

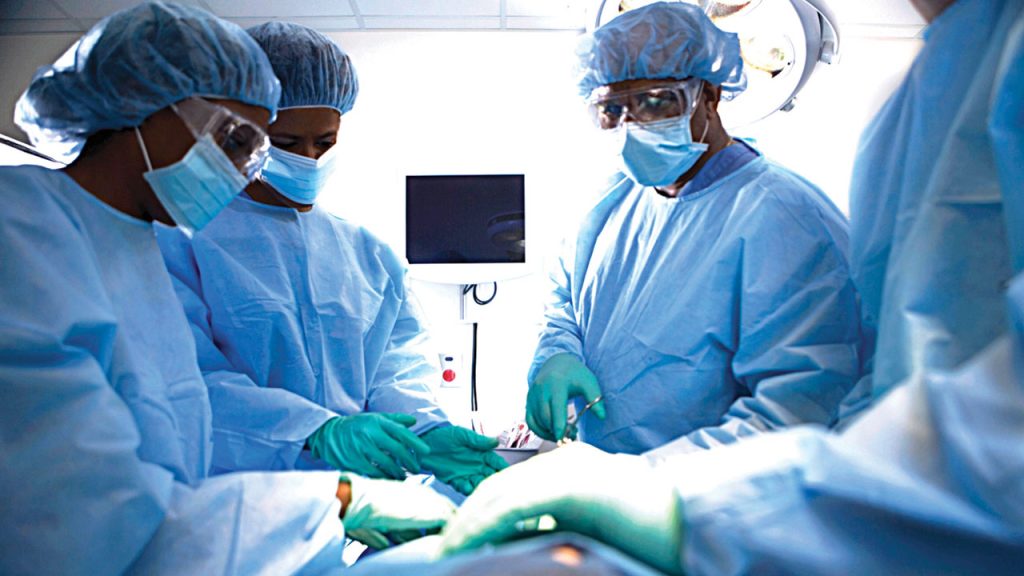

Why Selin Clinical Assessments Stands Out
At Selin Clinical Assessments, we prioritize accuracy, reliability, and efficiency in every clinical trial evaluation. Our experienced team ensures that each assessment is conducted with precision, providing researchers with the data they need to drive medical advancements. We’re committed to supporting groundbreaking research that leads to better treatments and improved healthcare outcomes.
We understand the importance of trust in clinical trials, which is why we adhere to the highest standards of quality and transparency. By utilizing advanced technology and proven methodologies, we ensure that all assessments are thorough, timely, and dependable, enabling research teams to focus on their core objectives.
Choosing Selin Clinical Assessments means partnering with a team that is dedicated to the success of your trial. Our commitment to excellence and our collaborative approach make us a reliable partner for organizations seeking accurate assessments to support their medical research initiatives.
Specialized Areas of Clinical Trial Expertise

Respiratory Trials

Rheumatology Trials

Infectious Disease Trials

Pediatrics Trials

Dermatology Trials

Women’s Health Trials
Core Values Driving Our Commitment to Excellence
At Selin Clinical Assessments, our core values drive every aspect of our work. We are committed to integrity in all our evaluations, ensuring that our assessments are accurate, transparent, and reliable. We believe that trust is fundamental to advancing medical research, and we uphold the highest ethical standards to foster that trust with our partners and clients.
Our dedication to innovation fuels our approach to clinical trials. We continuously seek out and implement the latest methodologies and technologies to enhance the accuracy and efficiency of our assessments. By staying at the forefront of industry developments, we support research that pushes boundaries and leads to meaningful medical breakthroughs.
Collaboration is central to our operations. We work closely with researchers, organizations, and stakeholders to understand their unique needs and provide tailored solutions. Our team values open communication and teamwork, believing that cooperative efforts lead to the most impactful outcomes in clinical research.
What Our Partners Are Saying
- Dr. Emily CarterSelin Clinical Assessments has been an invaluable partner in our research. Their precision and attention to detail have significantly contributed to the success of our trials.
- Dr. James NguyenThe team at Selin consistently delivers reliable and thorough assessments. Their commitment to quality is evident in every aspect of their work, making them a top choice for our clinical trials.
- Dr. Sarah PatelWorking with Selin Clinical Assessments has been a game-changer for our studies. Their innovative approach and dedication to accuracy have greatly enhanced our research outcomes.
- Dr. Michael RossSelin's professionalism and expertise have been instrumental in the success of our recent trials. Their ability to handle complex evaluations with ease is truly impressive.
- Dr. Olivia MartinezThe level of detail and thoroughness provided by Selin Clinical Assessments is unmatched. Their support has been crucial in advancing our clinical research projects.
- Dr. David KimSelin Clinical Assessments stands out for their exceptional service and reliability. Their team’s commitment to excellence has made a significant impact on our research efforts.
Let's Talk Now
Meet Our Expert Team

Dr. Michael Thompson
Expertise in evaluating new treatments for heart diseases and cardiovascular conditions.

Dr. Amanda Lee
Focused on evaluating new cancer treatments and innovative approaches in oncology research.

Dr. Robert Johnson
Specialist in assessing therapies for neurological disorders, including Alzheimer’s and Parkinson’s diseases.
